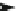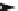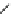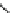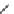Environmental Engineering Reference
In-Depth Information
4.4 Organic Oxidation Intermediates
Irrespective of fuel oil, fossil fuel or carbon-based fuel, oxidation intermediates
occur in any combustion reaction, because a combustion reaction cannot be con-
sidered complete even when oxygen is in excess or the flame temperature is high
[
20
-
22
].
As fuel combustion can be a longer or shorter process, one can assume that,
depending on the nature of fuel, it consists of the following phases that always end
in carbon dioxide formation [
20
].
4.4.1
Intermediate Oxidation Phases in Methane
Combustion (Zero Carbon)
0
−4
−2
+4
[O]
[O]
[O]
+2
[O]
CH
4
H
3
COH
H
2
CO H COOH
OCO
ð
4
:
24
Þ
4.4.2
Intermediate Oxidation Phases in the Combustion
of Primary Carbon Compounds
−3
−1
+3
[O]
[O]
+1
[O]
ð
4
:
25
Þ
RCH
3
RCH
2
OH
RCHO RCOOH
4.4.3
Intermediate Oxidation Phases in the Combustion
of Secondary Carbon Compounds
R
R
R
[O]
0
[O]
+2
CH
2
ð
4
:
26
Þ
CH
OH
C
O
R
R
R
4.4.4
Intermediate Oxidation Phases in the Combustion
of Tertiary Carbon Compounds
R
R
−1
[O]
+1
ð
4
:
27
Þ
R
CH
R
COH
R
R