Geology Reference
In-Depth Information
Functional group
A peripheral group of atoms
attached to the basic -C-C-C- skeleton of an
organic molecule, that bestows a particular chemi-
cal functionality.
waveform with a different shape and energy struc-
ture, e.g. the tetrahedral sp
3
hybrid characteristic of
many carbon compounds (Figures 7.5a and d) by
combining one 2s and three 2p orbitals.
Hydration
(a) Incorporation of water or OH
−
, cf.
dehydration
. (b) The stabilization of an ion in aque-
ous solution by the gathering of polar water mole-
cules around it. See Box 4.1.
Hydrothermal
Refers to hot saline aqueous fluids
circulating in the crust, or to veins and ore deposits
crystallized from such fluids.
Hydroxyl
Gas constant
R
A fundamental physical constant.
R
= 8.314 J mol
−1
K
−1
.
Geothermometry
Science of estimation of a rock's
temperature of formation (the temperature at which
its mineral assemblage equilibrated) from the com-
positions of the equilibrated minerals.
Glass
A state of matter having the disordered struc-
ture of a melt but the mechanical properties of a
solid.
The OH group or OH
−
ion.
Ideal solution
A solution sufficiently
dilute
for ion-
ion interactions to be insignificant.
Idiochromatic
Colour of a crystal that is derived
from one of its major elements (Gr.
idios
= own).
Incompatible element
See Box 9.1.
Incongruent melting
Melting of a mineral where a
new mineral is formed in addition to melt. See Box 2.6.
Inert gas
The family of gaseous elements helium (He),
neon (Ne), argon (Ar), krypton (Kr), xenon (Xe) and
radon (Rn) on the extreme right of the Periodic Table.
Infinitesimal
Half-life
t
1/2
The characteristic time it takes for a
radioisotope to decay to
half
its original concentra-
tion or radioactivity.
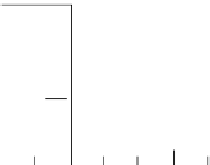
N
N
o
Readings of
N
taken at
different times
Describes something that is extremely
small.
Integral
The graph shows
y
varying as a function of
x
. The
integral
of
y
is an algebraic function express-
ing the area beneath the
y
curve between any speci-
fied limits
x
1
and
x
2
. It is written
∫
1
0.5
N
o
t
½
x
x
y
d
x
.
0
50
y
t
/hours
Hard water, hardness
Describes fresh water with a
relatively high natural content of Ca
2+
and Mg
2+
leached from aquifer minerals (usually carbonates)
by the reverse of reaction 4.2.1 (Box 4.2). Hardness is
the molar concentration of Mg
2+
+ Ca
2+
in mol l
−1
.
Heterogeneous (of a reaction or equilibrium)
Involving the participation of more than one chemi-
cal
phase
(e.g. reaction 4.12), cf.
homogeneous
.
Homogeneous (of a reaction or equilibrium)
Taking
place within a single chemical
phase
(e.g. reaction
3.1), cf.
heterogeneous
.
Hybrid, -ization
(of electron orbitals in atoms) The
merging of the wave functions of several valence
electrons in an atom to generate a multi-electron
x
1
x
2
x
Integrate, -ation
The mathematical process of calcu-
lating the
integral
of, or the area under, a curve.
Integration can be considered the reverse of differen-
tiation: thus the integral of velocity as a function of
time indicates the distance travelled.
Intensive
(of a variable) Having a magnitude inde-
pendent of the size of the system considered (e.g.
temperature).
Interfere, -ence
Term in optics describing interac-
tions between superposed waves (e.g. light rays) of



















Search WWH ::

Custom Search