Geology Reference
In-Depth Information
(a)
Pole
Snowfall on
polar icecap
Temperate
rainfall
Ocean
Evaporation
mainly in tropics
Equator
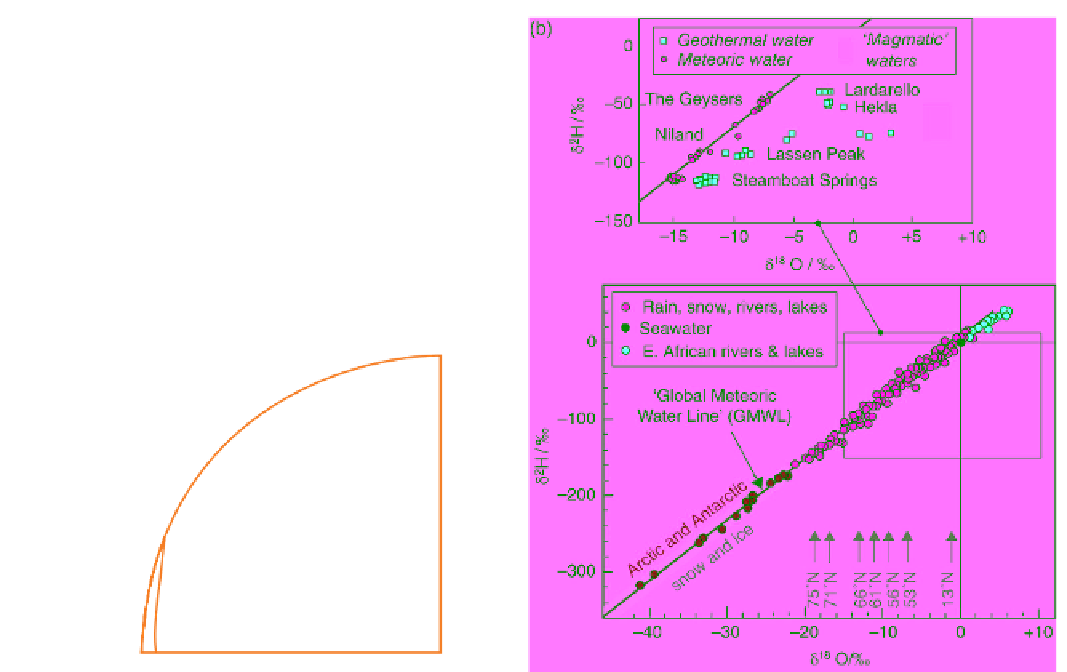
Figure 10.10
(a) Cartoon explaining why rain and snow become progressively more depleted in ΗDO and H
2
18
O with
increasing latitude; downward arrows represent precipitation. (b)
δ
D and
δ
18
O correlation in rivers, lakes, rain and snow,
after Craig (1961). Upward arrows illustrate latitude variation in
δ
18
O (after Dansgaard, 1964). The enlarged panel (after
Craig, 1963) shows how interaction with silicate rocks drives geothermal waters to higher
δ
18
O values while leaving the
δ
D
relatively unchanged, close to that of local meteoric waters from which the geothermal water has evidently been derived.
The field of 'magmatic waters' is from Taylor (1974).
(c) Geothermal waters, which are usually meteoric
waters that have equilibrated isotopically at high
temperatures with silicate rocks. Silicates (in which
oxygen is an essential constituent) constitute the
dominant oxygen reservoir of the Earth's crust.
Waters undergoing high-
T
isotopic exchange with
silicates tend to assimilate their positive
δ
18
O val-
ues. Silicate rocks contain little hydrogen, on the
other hand, so geothermal waters commonly retain
the latitude-related, negative
δ
D of the local met-
eoric waters from which they are derived. For this
reason, many geothermal waters define subhor-
izontal arrays of increasing
δ
18
O lying to the right
of the meteoric trend (Figure 10.10b, inset).
the water leaves its isotopic imprint on the rocks
affected by that process. Stable isotope investigation
of minerals in continental hydrothermal ore deposits,
for example, often reveals negative
δ
18
O values, point-
ing to a meteoric origin for the hydrothermal fluids
involved.
Stable-isotope palaeothermometry
and palaeoclimatology
A calcite crystal growing in equilibrium with seawater
at 25 °C is slightly enriched in
18
O relative to the water.
This can be expressed as a
fractionation factor α
(a kind
of
equilibrium constant
):
=
(
)
18
16
OO
OO
/
/
(10.9)
α
calcitewater
calcite
When large volumes of water are involved in a
geological process, especially at elevated temperatures,
(
)
/
18
16
water








Search WWH ::

Custom Search