Geology Reference
In-Depth Information
Energy
is simply the term that we use to describe a
system's
capacity for doing work
. Just as we recognize dif-
ferent forms of work (mechanical, electrical, chemical …),
so energy exists in a number of alternative forms, as will
be illustrated in the following pages. the energy stored in
an electrical battery, for example, represents the amount
of work that it can generate before becoming exhausted.
a system's capacity for doing work is necessarily expressed
in the units of work (just as the capacity of a bucket is
expressed as the number of litres of water it can contain),
so it follows that energy is also expressed in
joules = kg m
2
s
−1
. When discussing large amounts of
energy, we use larger units such as kilojoules (kJ = 10
3
J)
or megajoules (MJ = 10
6
J).
m
g
3
m
2
h
m
h
h
m
(b)
(d)
(a)
(c)
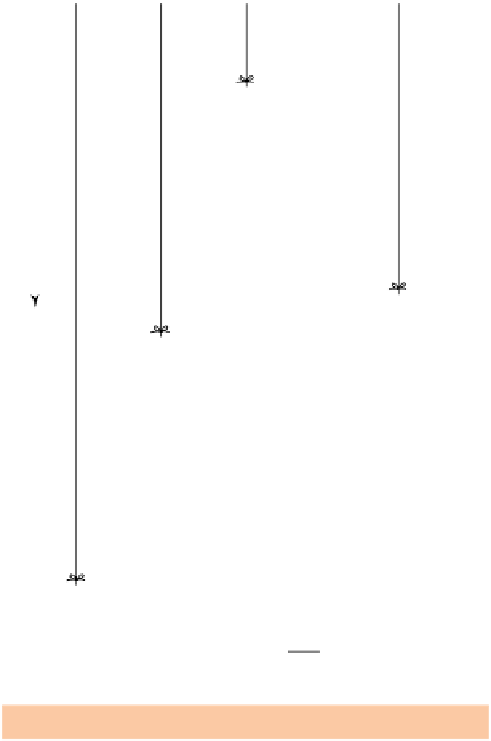
Figure 1.1.1
Work done in raising an object: (a) an object of
mass
m
resting on the ground; (b) the same object elevated
to height
h
; (c) the object elevated to height 2
h
; (d) another
object of mass 3
m
elevated to height
h
.
Note
: elevation is
measured between each object's
centre of gravity
in its initial
and final positions (note the centre of gravity of the larger
weight is slightly higher than the smaller one).
This simple example illustrates how one can go
about documenting the energy changes that accom-
pany geological reactions and processes, as a means of
understanding why and when those reactions occur.
This is the purpose of
thermodynamics
, a science that
documents and explains quantitatively the energy
changes in natural processes, just as economics anal-
yses the exchange of money in international trade.
Thermodynamics provides a fundamental theoretical
framework for documenting and interpreting energy
changes in processes of all kinds, not only in geology
but in a host of other scientific disciplines ranging from
chemical engineering to cosmology.
Thermodynamics, because it deals with very abstract
concepts, has acquired an aura of impenetrability in
the eyes of many Earth science students, particularly
those less at home in the realm of mathematics. With
this in mind, one objective of these opening chapters























Search WWH ::

Custom Search