Geology Reference
In-Depth Information
(a)
K shell
H
He
New Shell (L)
p sub-shell
New Shell (M)
Be
p sub-shell
Ne
Li
BC
N
O
F
Mg
Si
P
S
Cl
Ar
K
Ca
Na
Al
5
10
Atomic number
Z
15
20
(b)
Main diagram
2.5
He
New shells occupied
at Li, Na and K
1
Kr
Xe
(Rn)
Ne
0
50
90
Li Na K
b
s
(Fr)
2.0
Double occupation of p-orbital
lowers ionization energy
(mutual repulsion between
electrons in same orbital)
F
1. 5
Ar
N
H
O
Cl
C
1. 0
P
Be
S
Mg
B
Si
Ca
0.5
Na
First electron
enters p sub-shell
Al
Li
K
1
5
10
Atomic number
Z
15
20
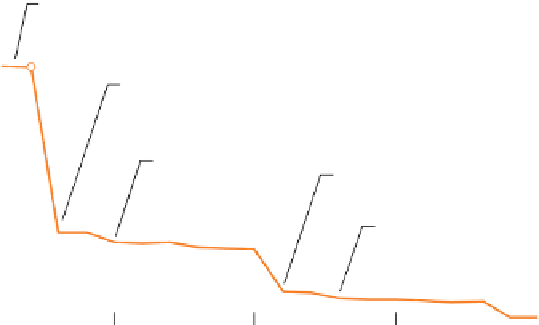
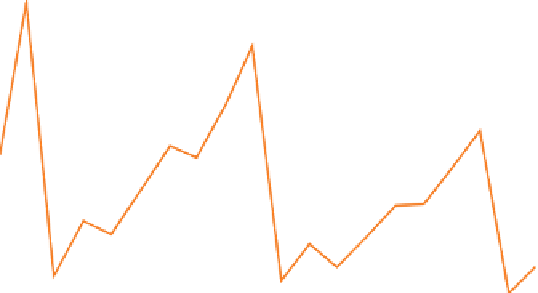
Figure 6.1
(a) A notional plot of ionization energy against atomic number, predicted without regard to the effect of increasing
nuclear charge. (b) The variation of
measured
ionization energy with atomic number
Z
among the first 20 elements. (The
whole
Z
-range is shown in the inset.) The rising profile between each abrupt drop reflects increasing nuclear charge.
by this single
valence electron
. All are
monovalent
metals: removing a second electron would be much
more difficult, because to do so would mean breaking
into the stable noble-gas core (Chapter 5). Because of
their low ionization energies, the alkali metals readily
form singly charged M
+
cations.
Beryllium (Be; 1s
2
2s
2
), magnesium (Mg; 1s
2
2s
2
2p
6
3s
2
)
and calcium (Ca; ls
2
2s
2
2p
6
3s
2
3p
6
4s
2
) all have two elec-
trons in their valence shells, both of which are fairly
easy to remove (although not as easy as the single elec-
tron in the valence shell of an alkali metal). These
alka-
line earth metals
utilize both of these electrons in their









































Search WWH ::

Custom Search