Environmental Engineering Reference
In-Depth Information
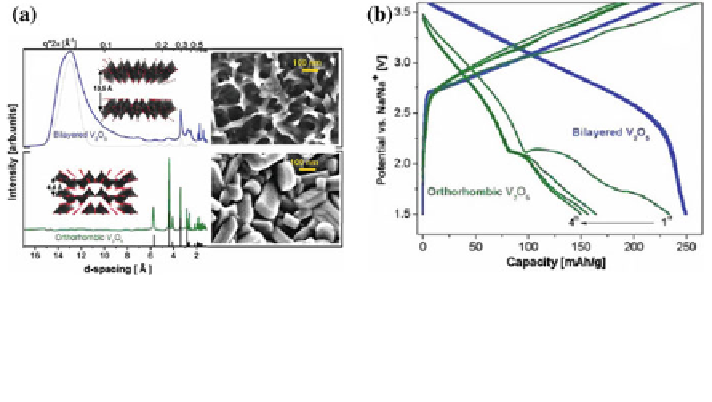
Fig. 8 a Synchrotron X-ray diffraction, scanning electron microscopy, and molecular simula-
tions of electrodeposited vanadium oxide: a bilayered V
2
O
5
annealed in vacuum at 120 C(blue);
b orthorhombic V
2
O
5
annealed in oxygen at 500 C(green). b First four charge/discharge cycles
of bilayered V
2
O
5
and orthorhombic V
2
O
5
electrodes. Both cells were cycled at 20 mA g
-1
,
within the potential window of 3.8-1.5 V (vs. Na/Na
+
) from 1 M NaClO
4
in PC solution [
16
]
solid-state reaction method can only accommodate 0.17 mole of Na
(~20 mAh g
-1
). The better performance of the material prepared by the sol-gel
process probably resulted from its high electrical conductivity along the b-axis,
high structural anisotropy and small particular size, which is favorable to fast
electronic and ionic diffusion [
15
]. V
2
O
5
with a bilayered structure and an
orthorhombic nanostructure were investigated [
16
]. XRD results showed that the
bilayered V
2
O
5
is composed of a stacking of V
2
O
5
bilayers based on the square-
pyramidal VO
5
units arranged in parallel (Fig.
8
a). The spacing distance of the
bilayers is approximately 13.5 Å, significantly larger than that (only 4.4 Å) of the
orthorhombic V
2
O
5
. Undoubtedly, the larger spacing would be more flexible to
accommodate the volume change due to the intercalation of Na ions. As shown in
Fig.
8
b, the bilayered V
2
O
5
electrode delivered a reversible capacity of
250 mAh g
-1
at 20 mA g
-1
, higher than that (only 150 mAh g
-1
) of the ortho-
rhombic V
2
O
5
. Besides, the bilayered V
2
O
5
electrode demonstrated higher average
discharge potential and reversible capacity on repeated cycling than its ortho-
rhombic counterpart. This investigation illustrated that tailoring the nanoarchi-
tecture of the materials can offer special functional properties to facilitate the
reversible insertion of Na ions.
Analogous to Na
x
CoO
2
and Na
x
MnO
2
,Na
x
VO
2
also has different lamellar
structure depending on the concentration of Na. The lamellar structure is com-
posed of VO
6
octahedra sharing edges to form VO
2
layers, which are stacked to
form O3 and P2-type structures according to the occupation sites of Na ions
(octahedral site (O3) for x = 1 and trigonal prismatic site (P2) for x = 0.7)
(Fig.
3
a, b). The electrochemical behaviors of O3-NaVO
2
and P2-Na
0.7
VO
2
have
been studied [
17
]. Both electrodes exhibited similar charge/discharge profiles
(Fig.
9
). The intercalation concentration of Na ion for both materials is up to at
least 0.5 mol of Na
+
per mole of NaVO
2
, corresponding to a reversible capacity of
Search WWH ::

Custom Search