Environmental Engineering Reference
In-Depth Information
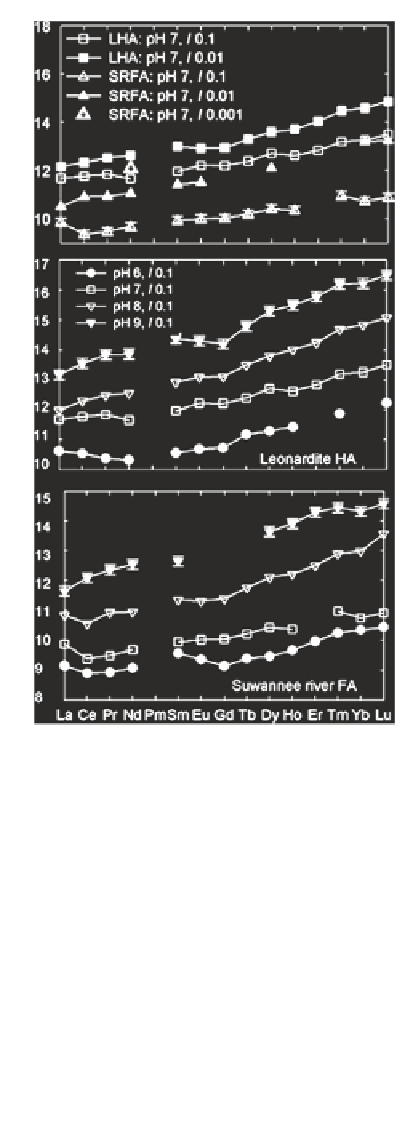
Fig. 5
Conditional
binding constants (log
K
)
for rare earth elements
(REE)-Leonardite coal
humic acid standard (LHA)
and REE-SRFA complex
formation at pH 7 with
various ionic strength (
a
) and
the effect pH on log
K
for
complexation of LHA (
b
) and
SRFA (
c
) with REE. Error
bars (0.16 log units) are only
shown for pH 7 (
a
), but apply
to all log
K
c values. Nominal
total concentrations are 100
nM REE; 20 mg L
−
1
SRFA,
130 nM EDTA for SRFA
experiments (
b
and
c
).
Data
source
Sonke and Salters
(
2006
)
(a)
(b)
(c)
Rare earth elements
Fig. 6
Log
K
for lanthanides and organic
ligands of various sources at pH 6, 7, and
8 as a function of ionic radius (sixfold
hydrated), including Sc and Y transi-
tion metals. Log
K
for Y and Leonardite
coal humic acid standard (LHA) values
are represented by singular data points,
floating just below the main rare earth ele-
ments (REE) data trends at an ionic radius
of 0.900 Å. Log
K
for lanthanides and
Elliot soil humic acid standard (EHA) val-
ues observes at pH 9, and 0.01 mol L
−
1
I
and extrapolated log
K
values for SRFA
and LHA under those conditions. EDTA
conditional binding constants for 0.01 mol
L
−
1
I
are included for comparison.
Data
source
Sonke and Salters (
2006
)
Ionic radius (angstrom)