Environmental Engineering Reference
In-Depth Information
THERMOCHEMICAL HYDROGEN PRODUCTION METHODS
STEAM REFORMING
PARTIAL OXIDATION
Natural Gas or
Light Hydrocarbon
Natural Gas or
Liquid Fuel
Oxygen
or air
Steam
Steam
Reformer
Partial
Oxidation
Heat
Syngas
H
2
, CO, CO
2,
CH
4
, H
2
O
Syngas
H
2
, CO, CO
2,
CH
4
, H
2
O
Shift Reactor
CO+H
2
O
→
CO
2
+H
2
Shift Reactor
CO+H
2
O
→
CO
2
+H
2
Steam
Steam
Waste
gases
Hydrogen
Purification
Waste
gases
Hydrogen
Purification
Pure H
2
Pure H
2
H
2
Storage
H
2
Storage
Compressor
Compressor
to H
2
Users
to H
2
Users
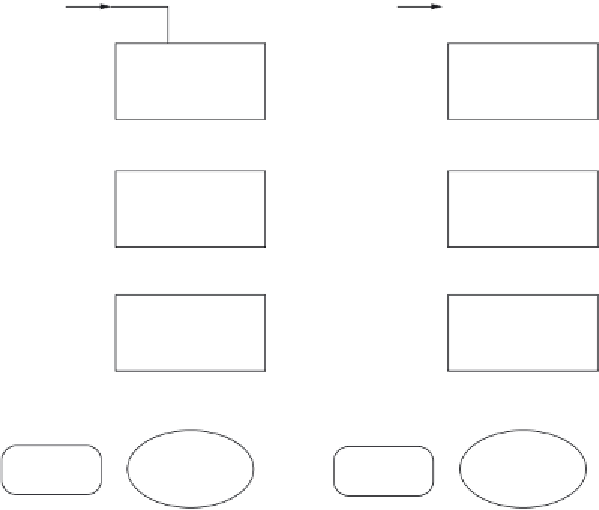
FIGURE 2.3
Comparison of the steam reforming and partial oxidation methods.
Source
: Reproduced
with permission from Ogden [1].
2.4 METHANOL AND ETHANOL STEAM REFORMING
Besides methane, methanol is another major candidate for H
2
production via
steam reforming through the following reaction:
CH OH H O CO
+
→
+
3
H
∆
H
=+
49
kJ mol
⋅
-
.
1
(2.5)
3
2
2
2
Among the different possible reactions involving methanol for hydrogen
generation, this reaction offers the highest maximum hydrogen content in
































Search WWH ::

Custom Search