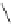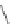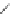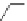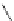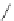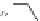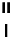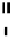Chemistry Reference
In-Depth Information
Table 2.11
Solution and gas phase basicity data for phosphazenes [90,91]
NMe
2
Me
2
NNP
NMe
2
NMe
2
NP
NMe
2
NMe
2
N
t
Bu
NR
2
R
2
NNP
N
NR'
NR
2
NR
2
NR'
NR
2
P
NMe
2
P
NR
2
NP
NR
2
NR
2
R
2
N
P
NR
2
N
P
NR''
2
N
P
NR
2
NR
2
R
2
N
P
NR
2
NR
2
101
R = Me
NR
2
= pyrr
R'P
3
(dma)R''
R'P
3
(pyrr)R''
102
: R = Me; R' =
t
Bu
103
: R = Me; R' = CMe
2
CH
2
t
Bu
104
: R = Me; R' = CEt
3
105
: R = Me; R'= CMe
2
t
Bu
106
: NR
2
= pyrr ; R' =
t
Bu
107
: NR
2
= pyrr ; R' = 2-ClC
6
H
4
108
: R = R'' = Me, R' =
t
Bu
109
: R = Me, R'' =
i
Pr, R '=
t
Bu
NR
2
R
2
NNP
N
NR'
NR
2
NR
2
P
NR
2
NP
NR
2
N
P
NR
2
NR
2
NMe
2
Me
2
NN
NMe
2
N
N
t
Bu
NP
NMe
2
NMe
2
N
R
2
N
P
NR
2
NR
2
NMe
2
NMe
2
NMe
2
P
NMe
2
P
NMe
2
P
N
P
110
: R = Me; R' =
t
Bu
111
: NR
2
= pyrr ; R' =
t
Bu
NR
2
R
2
NNP
NR
2
NR
2
Me
2
N
PNMe
2
P
NR
2
NR'
N
P
NMe
2
Me
2
N
NMe
2
N
t
Bu
NP
NMe
2
NMe
2
NMe
2
NNP
N
i
Pr
2
NMe
2
NP
NMe
2
NMe
2
NMe
2
112
: R = R' = Me
113
: R = Me, R' = Et
114
: R = Me, R' =
t
Bu
115
: R = Me, R' = cctyl
116
: NR
2
=pyrr, R' =
t
Bu
R'P
2
(dma)
Me
2
N
P
NMe
2
P
NMe
2
118
117
R'P
2
(pyrr)
NR'
Me
R
Me
R
2
N
P
2
N
NMe
2
Me
2
NNP
N
N
N
N
NR
2
Et
2
NP
N
N
P
P
NMe
2
N
Me
Me
119
: R = Me; R '=
t
Bu
120
: R = R' = Me
121
: NR
2
= pyrr ; R' =
t
Bu
N
N
R'P
1
(dma)
t
Bu
t
Bu
Et Me
R'P
1
(pyrr)
122
125
: BEMP
123
: R = Me;
124
: R = Pr
p
K
a
DMSO
(H
2
O) GB
exp
GB
calc
p
K
a
calc
(MeCN)
p
K
a
(THF)
Base
p
K
a
(MeCN)
111
P
5
46.9
118
P
7
(45.3)
>
32
110
P
5
45.3
>
32
117
P
5
(44.0)
106
P
4
44.0
105
P
4
(42.7)
104
P
4
(42.7)
103
P
4
(42.7)
29.4
102
P
4
(42.7)
30.2
108
P
3
38.6
26.2
109
P
3
38.6
26.1
4-MeO-C
6
H
4
P
4
(pyrr)
28.9 [76]
101
P
4
36.6
114
P
2
33.49
21.5
107
EtP
3
(dma)
25.3
115
P
2
33.27
124
P
2
33.11
123
P
2
33.08
113
P
2
32.94
21.1
112
P
2
32.72
(continued)