Biomedical Engineering Reference
In-Depth Information
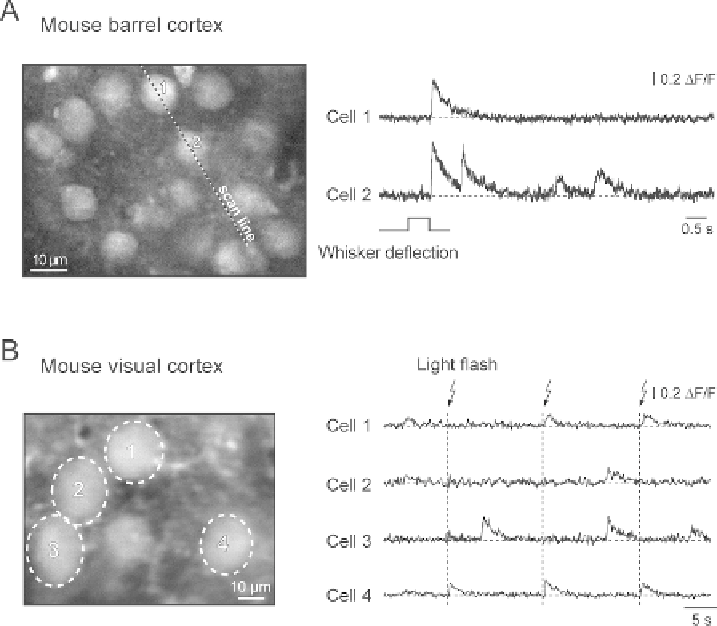
Fig. 3.7. Sensory-driven Ca
2
+
transients in individual cortical neurons. A, individual neurons in the mouse barrel cortex
(left) and the corresponding Ca
2
+
transients (right) evoked by whisker deflection. Whiskers at the contralateral side of
the snout were moved by air puffs. The transients were recorded using the line-scan mode (5 ms/line; the position of the
scanned line is indicated). B, individual neurons in the mouse visual cortex (left) and the corresponding Ca
2
+
transients
(right) evoked by brief consecutive light flashes. A is reproduced, with permission from National Academy of Sciences
USA, copyright 2003, from Stosiek et al.
(38)
, B is reproduced, with permission from Springer-Verlag copyright 2006
from Garaschuk et al.
(112)
.
the dorsal surface of the olfactory bulb. This area encompassed
approximately 150 glomeruli. While functional imaging meth-
ods such as fMRI, 2-deoxyglucose, and intrinsic imaging reveal
patterns of glomerular activity (e.g.,
(47-49)
), with these meth-
ods, it is difficult to determine whether the signals come from
presynaptic terminals and/or from post-synaptic juxtaglomeru-
lar and/or mitral/tufted processes in the glomerulus. In selected
instances, labeling neurons with voltage- or calcium-sensitive dyes
via retrograde or anterograde transport allows selective monitor-
ing of activity in defined neuronal populations (e.g.,
(50-52)
).
This approach was first developed for the olfactory receptor neu-
rons by Friedrich and Korsching
(53)
in the zebrafish and later
adapted for use in the mouse
(54)
.
We tested one calcium-sensitive dye, Calcium Green-1
(Invitrogen-Molecular Probes, Eugene OR), that was not
dextran conjugated. While it did label receptor neurons in the
2.3.1. Dye Screening