Environmental Engineering Reference
In-Depth Information
are very similar: g
iso
¼ 2.02 (A
iso
,
Co
¼ 12 - 14 G), g
k
¼ 2.09, g
?
¼ 2.00 (A
k
¼ 15 - 17
G) [LeMest et al., 1997]. Certainly, further work is required to use this EPR data to
quantify the distribution of electron density in these adducts.
In benzonitrile, doubly oxidized Co
2
group 2 porphyrins also bind O
2
, albeit more
weakly than the singly oxidized analogs [LeMest et al., 1997]. The electronic con-
figuration of the resulting adducts, [(dipor)Co
2
O
2
]
2
þ
, is poorly understood.
[(FTF4)Co
2
O
2
]
2
þ
was reported [LeMest et al., 1997] to give a single-line EPR spec-
trum indicative of a p-cation radical. The [(dipor)Co
2
O
2
]
2
þ
adduct is an even-electron
system and must have two unpaired noncoupled electrons to give an EPR signal of an
organic radical. The corresponding electronic state is unknown.
Stepwise oxidation of [(FTF4)Co
2
O
2
] to the mono- and dications was reported to
proceed reversibly in benzonitrile [LeMest et al., 1997]: such reversible redox chem-
istry has not been reported for any O
2
adducts of monomeric Co or Fe porphyrins.
There is a major controversy over which bridging mode (Fig. 18.14) the O
2
moiety
adopts in the [(dipor)Co
2
O
2
]
þ
adducts. Among the three conceivable modes, the
m-1,2 O
2
ligand is by far the most common in transition metal/O
2
chemistry, and is
quite flexible with respect to the M - M separation. For example, the Co
...
Co distance
in the Co - O - O - Co moiety in crystallographically characterized complexes varies
between 4.9 and 2.8
˚
. The wide range of M - M distances seen in crystal structures
of derivatives of pillared cofacial porphyrins suggests that the diporphyrin core may
also accommodate a range of Co
...
Co distances, including those optimal for the O
2
ligand coordinating in the m-1,2 mode. On the other hand, the m, h
2
: h
2
binding
mode is much less common, and is less flexible, imposing restrictions on the extent
of lateral displacement of the two porphyrin rings that are exceeded by those seen
in the crystal structures of most pillared porphyrins.
Resonance Raman studies of dioxygenated cofacial porphyrins similar to
(FTF4)Co
2
and of [(DPA)Co
2
O
2
] [Proniewicz et al., 1989] failed to differentiate the
binding modes. The two adducts manifested similar O - O stretching frequencies
n(O
2
), 1081 and 1085 cm
21
, despite the different solid state stereochemistry of the
parent (deoxygenated) complexes. The n(O
2
) values are comparable to those in
doubly bridged [(NH
3
)
8
Co
2
(m-O
2
)(m-NH
2
)]
4
þ
adducts containing a trans-m,h
1
: h
1
-O
2
2
Three possible modes of O
2
binding in [(dipor)Co
2
]
x
þ
(x ¼ 0, 1, 2).
Figure 18.14
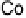
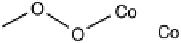
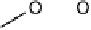
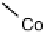


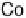
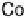
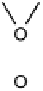
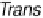

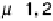
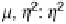
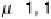











Search WWH ::

Custom Search