Environmental Engineering Reference
In-Depth Information
This would be consistent with a transformation of a CO
ad
-rich (2
2)(2CO
þ
O)
adsorbate phase [Schiffer et al., 1997] into an oxygen-rich, but still CO
ad
-containing
phase, for example the (2
2)(CO
þ
2O) phase known from UHV experiments
[Narloch et al., 1994]. CO adsorption on the Ru(0001) surface at 0.7 V is essentially
inhibited [Wang et al., 2001], most likely as a result of surface blocking by OH
ad
/
O
ad
species.
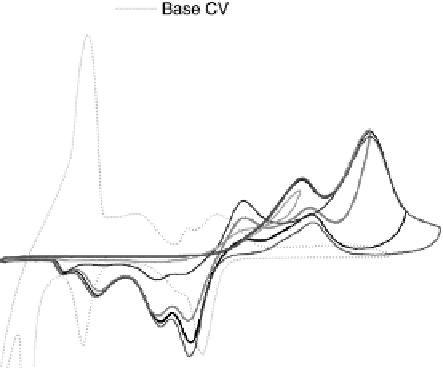
Potentiodynamic CO bulk oxidation on a nonmodified Ru(0001) electrode is
shown in Fig. 14.9 (Plate 14.1) (solid lines), including cycles with increasing
anodic limit. For comparison, a base CV (dotted line) is included as well (as in
Fig. 14.2, but at a scan rate of 10 mV s
21
). Only for E . 0.55 V are anodic currents
observed in a CO-containing electrolyte in the positive-going and (less pronounced)
also in the negative-going potential scan, indicative of continuous CO oxidation. In
the negative-going scan, we find reduction charges at potentials 0.55 V . E . 0.1
V. They grow for higher anodic potential limits, and finally approach a value of
about 0.5 mC cm
22
at an anodic limit
1.2 V. This is the charge expected for the
reduction of 1 ML O
ad
to H
2
O, in good agreement with our assignment of a (1
1)O adlayer on Ru(0001) at E . 1.05 V in a CO-free electrolyte (see above). In
CO-free electrolyte, this reduction takes place in the peaks C
0
,B
0
, and part of A
0
,
whereas in the presence of CO, the voltammetric pattern is totally different, even
for E , 0.55 V, where CO oxidation does not occur. We explain this difference by
the gain in energy due to adsorption of strongly adsorbing CO in a CO-containing sol-
ution, compared with O
ad
reduction and the OH
ad
!
H
upd
exchange in the absence of
Figure 14.9 CO bulk electro-oxidation at bare Ru(0001) in flow cell; dotted line, CO free
electrolyte; solid lines: flow of CO saturated electrolyte, with varied upper scan limits (see
key on figure). (See color insert.)





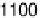


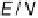






















Search WWH ::

Custom Search