Agriculture Reference
In-Depth Information
2008; Figure 2.2). It is not possible to differentiate GH 31 substrate specificities
from genome sequences (Benson et al., 2008). Both enzymes are internally dupli-
cated, have two catalytic sites, and are bound to the enterocyte membrane at the
N-terminal (Quezada-Calvillo et al., 2007a, 2007b). MGAM and SI both have two
aspartic acid proton donors and aspartic acid nucleophiles in a (β/α)
8
barrel struc-
ture. Both enzymes are exoglucosidases that can hydrolyze granular starch, but the
rate of glucose production is amplified fivefold if starch is also treated by AMY. The
rate of α-glucogenesis from maltodextrins and α-limit dextrins by MGAM is tenfold
greater than SI, but MGAM is inhibited by lumenal substrates and SI is not. This
suggests that although sharing the same substrates, MGAM accelerates glucogen-
esis on low-starch diets, and SI constrains glucogenesis after higher starch intakes
(Quezada-Calvillo et al., 2007b).
The concept of glycemic index (GI) of foods was introduced in the 1980s (Englyst
et al., 2007). The GI is calculated by comparing the rise of blood glucose of fasting
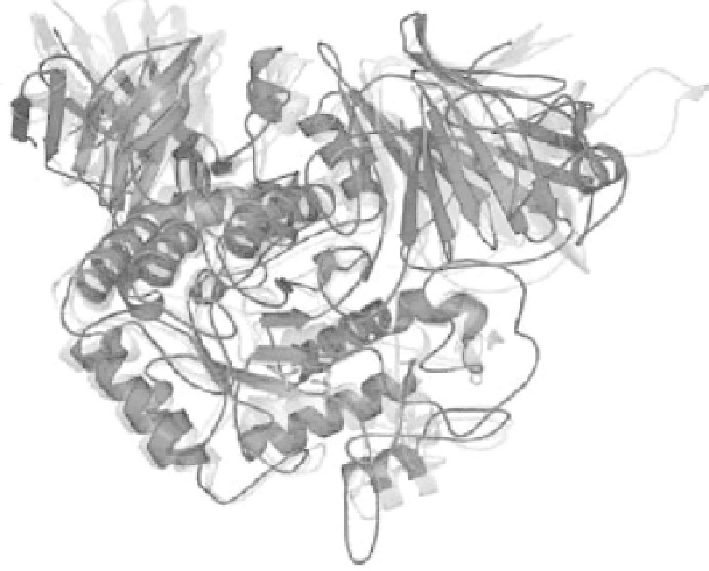
fIguRe 2.2
A color version of this figure follows page 198.
Phylogenetic conservation
of GH 31 enzyme sequences and structures. The superposition of the human N-terminal of
human maltase-glucoamylase (MGAM) (red), archael YicI (cyan), and bacterial MalA (yel-
low) structures is shown within a conserved (β/α)
8
barrel structure. Glycerol molecules that
indicate locations of glucose-binding areas in the active-site MGAM are represented as stick
figures. Note the conservation of the tertiary protein structures, which diverged over more
than 2.5 billion years. (From Sim, L., Quezada-Calvillo, R., Sterchi, E.E., Nichols, B.L., and
Rose, D.R.,
J Mol Biol
. 2008 Jan 18;375(3):782-792. With permission.)