Environmental Engineering Reference
In-Depth Information
been interchanged with respect to their positions in the emf series. To minimize
galvanic corrosion, the rule of thumb is to avoid the coupling of metals and alloys
far apart in the series.
The galvanic series in seawater is widely used to predict the galvanic behavior
in other environments as well. However, this should be done with caution.
Changes in electrolyte composition and temperature can significantly change the
electrode potential (see Nernst equation) and hence cause a change in position
in the galvanic series. Tin has been shown as nobler than iron in Table 3.1. In
''tin cans,'' however, certain food constituents combine chemically with Sn
2
ions to form soluble tin complexes. This lowers the activity of Sn
2
ions, shifting
the potential of tin to more active direction so that iron may become cathodic.
Ideally, there should be a galvanic series for each environment, but this is not
practicable because it would require an infinite number of tests. Even then it may
not help in a quantitative prediction of galvanic corrosion.
3.3.3 Sacrificial Anode
The fact that the cathodic member in the galvanic couple remains free from corro-
sion is utilized to protect a structure or component by making it cathode. This
is accomplished by coupling the structure or coating it with a less noble metal.
The anode protects the structure by sacrificing its life through preferential dissolu-
tion, hence the name ''sacrificial anode.'' A ship hull made of steel is protected
by insertion of magnesium blocks at places. Such a protection is referred to as
cathodic protection, which is discussed in detail in Section 4.4.
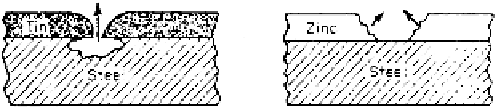
Galvanizing is a common protective measure for steel parts. A layer of zinc
is provided on steel by hot dipping in molten zinc. The layer is never perfect.
Also, local breakdown or cracking of the layer occurs due to mechanical damage.
At these discontinuities the corrosive comes in contact with both steel and zinc.
Zinc being the anode dissolves, leaving the steel intact. Although the anodic area
is large, the rate of dissolution is slow and this ensures a reasonable coating life
as well. Coating steel with a more noble metal, say tin, would have provided a
reverse situation of small anode (steel)-large cathode (tin) at the discontinuities,
inviting a rapid attack on steel (Fig. 3.4). However, it may be noted that in some
Figure 3.4
Galvanic corrosion at discontinuities in tin- and zinc-coated steel.