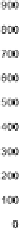Environmental Engineering Reference
In-Depth Information
Source:
IPCC, 1999, and Lee et al, 2002a
Figure 5.6
Development over time of fuel usage and NO
x
emissions from aviation
growth) between the Fa and Fe scenarios (mid- and upper-growth scenarios, respec-
tively), but an aggressive NO
x
reduction, resulting in a global NO
x
emission closer
to the Fc scenario (lowest growth of the FESG scenarios). For the DTI 2050 sce-
nario, the global fuel consumption was estimated to be 633Tg, with 4.43Tg of NO
x
(as NO
2
), produced as a consequence of a fleet emissions index (EI) of 7.
E
FFECTS
OF
SUBSONIC
AVIATION
Effects of NO
x
emissions from aircraft
Emissions of NO
x
arise from the combustion of aviation fuel (kerosene) and are a
complex function of combustion temperature, pressure and combustor design. These
emissions are primarily nitric oxide (NO) but are quickly converted to nitrogen
dioxide (NO
2
) in the atmosphere.
Emissions of NO
x
result in the catalytic production of tropospheric O
3
via a
number of complex chemical processes (for a review, see IPCC, 1999, Chapter 2).
Essentially, oxidation of methane (CH
4
), carbon monoxide (CO) and non-methane
hydrocarbons (NMHCs) results in the production of the hydroperoxy radical
(HO
2
). This HO
2
then reacts with NO to form NO
2
, which may subsequently pho-
todissociate, reforming NO and liberating the highly reactive atomic oxygen (O).
Atomic oxygen reacts with O
2
, forming O
3
. This chemical process occurs in the nat-
ural atmosphere, but the introduction of extra NO
x
catalytically enhances the pro-
duction rate of O
3
. This is summarized in Figure 5.7, in which the catalytic role of
NO
x
is shown, forming and recycling hydroxyl radicals (OH) and hydroperoxy rad-
icals (HO
2
).
The production of O
3
at altitudes at which subsonic aircraft typically fly (10-
12km) is rather efficient and it is not so readily removed from the atmosphere as it is
at the ground (by dry deposition), thus resulting in a residence time of weeks. More-
over, the temperature response for a change in O
3
is altitudinally dependent, as
shown in Figure 5.8. This is because O
3
absorbs both incoming solar radiation and
outgoing infrared radiation. At around the tropopause level, the sensitivity is great-
est as the radiative forcing is proportional to the difference in the temperatures at