Biology Reference
In-Depth Information
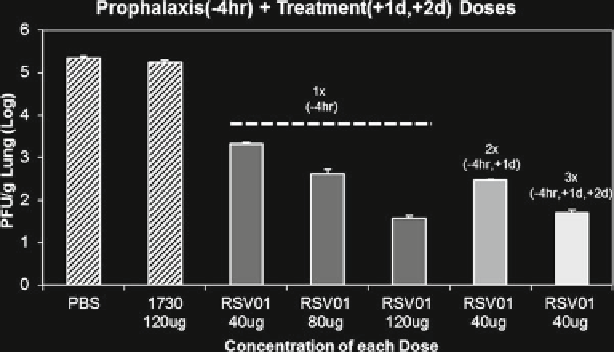
Fig. 15.1
In vivo activity of ALN-RSV01 in BALB/c mice. ALN-RSV01 dose-response curve of
mice treated with single doses of 40, 80, or 120 mg ALN-RSV01 intranasally 4 h prior to infection
or with multiple doses (as indicated) pre- and post-infection with 1 × 10
6
PFU of RSV A2. The
lungs were harvested, and the virus was quantified by a standard immunostaining plaque assay.
Each bar represents the mean and standard deviation of data from five animals. 1730 is a mismatch
siRNA control
15.2.2
In Vivo Studies of ALN-RSV01
The BALB/c mouse is a well-established model for RSV infection and was thus
chosen as the in vivo system for evaluating antiviral efficacy of ALN-RSV01.
Studies were initially performed in a prophylaxis model where the ALN-RSV01
was administered intranasally (i.n.) by pipette (25 ml per nostril) to mice 4 h prior to
infection with 10
6
pfu of RSV/A2. There was a dose-dependent inhibition of RSV/
A2 replication in the lungs of mice, with a 120 mg dose reducing RSV titers by >3.0
log
10
pfu/g lung as compared to either PBS or a nonspecific siRNA control (Fig.
15.1
).
In addition, ALN-RSV01 was equally efficacious when the same total siRNA dose
was delivered over two split doses (4 h prior and 1 day after) or three split doses (4 h
prior, 1 day and 2 days after) (Fig.
15.1
).
To evaluate the efficacy of viral inhibition in a treatment paradigm, ALN-RSV01
was delivered i.n., in single or multiple daily doses 1-3 days post-infection. When
ALN-RSV01 was administered in a treatment regimen as a single dose following
viral inoculation, dose-dependent antiviral efficacy was found to decrease as a func-
tion of time of dosing post viral infection: when multiple 40 mg doses of ALN-
RSV01 were delivered daily on days 1-3, potent antiviral activity was seen, and
viral titers were reduced to background levels, however, by day 3 postinfection;
single doses as high as 120 mg did not result in significant viral inhibition.
Search WWH ::

Custom Search