Environmental Engineering Reference
In-Depth Information
Figure 9.4 presents the typical magnetization loop for synthesized -Fe
2
O
3
,
where no reduced remanence and coercivity were observed. Saturation moment, M
s
,
indicative of the magnetic properties was reached at 9000 Oe and found to be 3.3 emu,
which is very similar to bulk values (3.4 emu) (Svoboda, 1987). It is known that
magnetic particles of less than 30 nm will exhibit superparamagnetism (Watson and
Cressey, 2000). This superparamagnetic property of the -Fe
2
O
3
nanoparticles makes
them very susceptible to the magnetic field, and therefore renders the nanoparticles to
separate easily from the solution. As found, these -Fe
2
O
3
nanoparticles can be
completely separated from the solution within 20s. Without the external field, the -
Fe
2
O
3
nanoparticles do not show any magnetic properties toward the surroundings. The
surface area of the freeze-dried -Fe
2
O
3
was measured and found to be 162 m
2
/g. The
point of zero charge pH
pzc
of -Fe
2
O
3
nanoparticles was determined to be approximately
6.3.
a
-
Fe
2
O
3
b
-
Fe
2
O
3
2
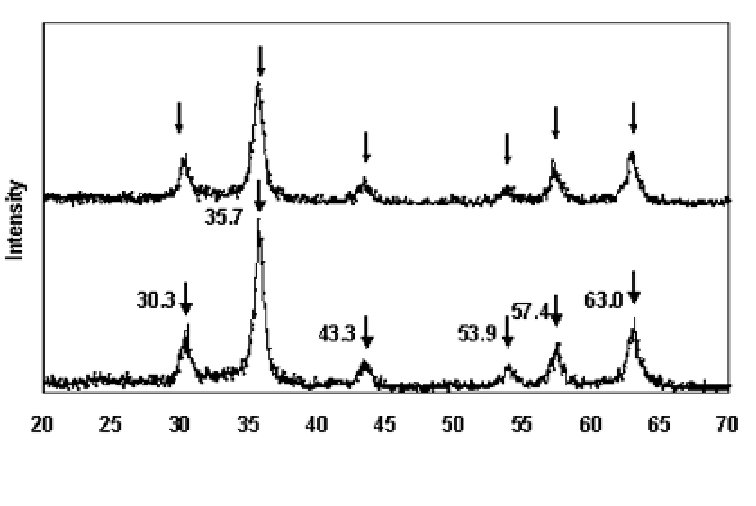
(degree)
Figure 9.3
XRD patterns of -Fe
2
O
3
(a) before and (b) after Cr(VI)
adsorption.
The -Fe
2
O
3
nanoparticles synthesized by high-temperature decomposition of
metal precursors were dispersed well in the hexane solvent as shown in Figure 9.5. The
surface area of the -Fe
2
O
3
nanoparticles of 3, 7, 11 nm is 342, 280, 246 m
2
/g,
respectively. Obviously, the surface area of the -Fe
2
O
3
nanoparticles increased with
decreasing particle size. The XRD profile presents the highly crystalline structure of

Search WWH ::

Custom Search