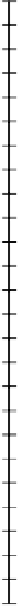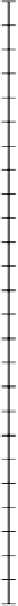Agriculture Reference
In-Depth Information
15
15
14
14
130
O
2
H
2
O
O
2
H
2
O
13
13
NO
3
−
N
2
10
12
120
12
11
11
20
NO
3
−
N
2
O
MnO
2
Mn
2
+
110
10
10
9
9
30
100
8
8
NO
2
−
NO
3
−
NO
3
−
NO
2
−
7
40
7
90
NO
3
−
NH
4
+
6
6
NH
4
+
NO
2
−
50
80
5
5
4
4
60
70
3
3
2
2
70
60
1
1
80
50
0
0
Fe(OH)
3
Fe
2
+
−
−
−
−
−
−
−
1
−
−
−
−
1
Fe
2
+
Fe(OH)
3
90
40
CH
2
O CH
3
OH
SO
4
2
−
HS
−
100
30
H
2
S SO
4
2
−
110
20
−
−
−
−
CO
2
CH
4
120
10
H
2
H
+
H
+
H
2
−
−
130
CH
2
O CO
2
−
1
−
−
10
Figure 4.3
Free energy changes in redox reactions mediated by microbes. (a) Oxidation of reduced inorganic compounds linked to reduction
of O
2
. (b) Oxidation of organic matter 'CH
2
O' linked to reduction of various organic and inorganic oxidants. pH
=
7 and unit oxidant and
reductant activities except
(
Mn
2
+
)
=
0
.
2mM and
(
Fe
2
+
)
=
1mM