Biology Reference
In-Depth Information
Physical titer
Functional titer
Elisa p24
qPCR / RNA titer
For all lentiviral
vectors
For all lentiviral
vectors
293T
Harvest
FACS
qPCR / DNA titer
For lentiviral vectors
with ubiquitous
promoters and VSV-G
or MOK-G envelope
For lentiviral vectors
with any promoters
and VSV-G or MOK-G
envelope
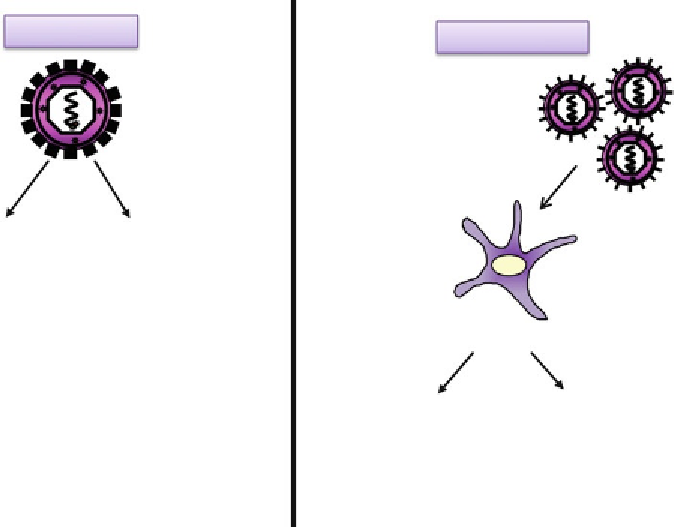
Fig. 7
Schematic diagram showing the various ways to titer lentiviral vectors. Physical titers can be obtained
by ELISA and the determination of p24 antigen or by RT-qPCR on viral suspensions. Functional titers can be
determined by cytometry or DNA titer determinations on infected cells [
108
]
19,000 rpm (Beckman Coulter SW 32Ti rotor) for 1.5 h at 4 °C.
The supernatant is removed and the pellets are pooled and resus-
pended in 1 ml of 1 % BSA in PBS. This viral suspension can be
used for in vitro experiments. For in vivo experiments, a second
concentration step is required (19,000 rpm, Beckman Coulter SW
48Ti rotor) (
see
Note 14
). The fi nal pellet, containing the LV, is
resuspended in 120
l of 1 % BSA in PBS and frozen at −80 °C
until use. When scaling up production, we resuspend the pellet in
300-400
μ
l of buffer per fi ve stacks. For LVs pseudotyped with the
MOK-G envelope or containing a miRT sequence with a lower
production yield, the pellet is resuspended in a smaller volume
(e.g., ~60
μ
μ
l).
Several methods can be used to determine functional or physical
lentiviral titers as shown in Fig.
7
. The advantage of the physical
titer is that it can be used for batches regardless of the promoter
used and independently of the presence or absence of miRT or the
envelope used for pseudotyping. It provides a quantitative estimate
of the total number of physical particles. This method is particu-
larly suitable for adjusting LV titer as described here. By contrast,
the functional titer provides a measurement of the transduction
3.7 LV Titration
Search WWH ::

Custom Search