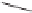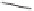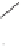Chemistry Reference
In-Depth Information
Tab l e 9 . 6
Yield (%)
Substrate
Product
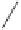
Ph
MeO
2
C
Ph
MeO
2
C
93
O
MeO
2
C
MeO
2
C
Ph
Ph
97
O
O
O
Bu
Bu
89
TsN
TsN
O
O
+
Ph
90
Ph
O
93
+
Cobalt-rhodium heterobimetallic nanoparticles were synthesized from Co
n
Rh
4-n
(CO)
12
and immobilized on charcoal. The method used in this study gave fixed stoichiometric
Co/Rh bimetallic nanoparticles.
Several years ago, Casanove reported
36
on the structure and chemical order in Co-Rh
(Co
x
Rh
1-
x
) nanoparticles. Their synthesis was based on the co-decomposition under di-
hydrogen (3 bar) of [Co(
4
-C
8
H
12
)], in a
solvent containing either a polymer or a ligand. Based on their study, a marked segregation
of cobalt atoms at the surface was proposed (Figure 9.4).
3
-C
8
H
13
)(
4
-C
8
H
12
)] and [Rh(acetylacetate)(
Figure 9.4
420-atom clusters after MCM simulated. Black balls: Co; White balls: Rh. From Left
to right: Co
0.25
Rh
0.75
,Co
0.5
Rh
0.5
,andCo
0.75
Rh
0.25
. Top: general view; bottom: cross-sections
[from ref. 36].