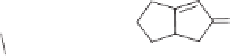Chemistry Reference
In-Depth Information
3 mol% [RhCl(CO)
2
]
2
6 mol% (
S
)-BINAP
12 mol% AgOTf
R
R
X
O
X
∗
CO (1 atm)
H
THF, 90
°
C, 3-5 h
Me
Ph
Ph
O
O
O
O
O
∗
∗
∗
H
H
H
Yield = 61%
Ee = 51%
Yield = 40%
Ee = 96%
Yield = 88%
Ee = 81%
Me
Me
i-
PrO
2
C
i-
PrO
2
C
O
O
TsN
∗
∗
H
Yield = 40%
Ee = 90%
H
Yield = 80%
Ee = 84%
Scheme 7.3
Rh-catalyzed Pauson-Khand reaction of 1,6-enynes.
They proposed a cationic pathway for the asymmetric PKR mechanism. 1,6-enyne was
suggested to bind with [Rh(CO)(
S
)-BINAP]
+
complex and transform to octahedral Rh(III)-
metallocyclopentene intermediate. Subsequent migratory insertion of CO and reductive
elimination furnished the desired carbonylative cycloadduct.
4
Based on the success of
the [RhCl(CO)
2
]
2
complex in intramolecular cycloaddition of 1,6-enynes, Narasaka
et al.
further extended its application to intermolecular Pauson-Khand-type cyclization.
5
They
reported that the reaction between norbornene and 1-phenylpropyne gave the desired cyclic
enones in 69% product yield as a mixture of regioisomers (Scheme 7.4). This Rh-catalyzed
reaction was accelerated by reducing CO partial pressure to less than 1 atm.
5 mol% [RhCl(CO)
2
]
2
O
O
+
Ph
Me
Bu
2
O, 130
°
C, 60 h
+
CO (1 atm)
Me
Ph
69% yield
(53:47)
Me
Ph
Scheme 7.4
Rh-catalyzed intermolecular PKR of norbornene and 1-phenylpropyne.
In 2003, Chung and co-workers established a recyclable Pauson-Khand-type protocol
assisted by the entrapped Rh complexes, which were prepared by a sol-gel process.
6
The
metal complex could be recovered and reused at least ten times without loss of catalytic
activity. However, the stereo-induction offered by this catalytic system was poor. Only 13%
ee was obtained in intramolecular Pauson-Khand-type cyclization of aromatic 1,6-enyne.