Chemistry Reference
In-Depth Information
3-carboxamido regioisomer
70
is exclusively formed; the
N
-(tetrolyl)oxazolidinones
69
give mixtures of 3-carboxamido (
70
) and 2-carboxamido (
71
) regioisomers. These regioi-
somers are readily separated by column chromatography. In general, the 3-carboxamido
regioisomers
70
are obtained with good diastereoselectivity, remarkably those derived
from commercially available 4-phenyloxazolidinone. These diastereomers are also read-
ily separated by column chromatography, and in general the major diastereomers of
70
can be isolated in stereochemically pure form in reasonable yields. On the other hand,
2-carboxamido regioisomers
71
are formed with much less diastereoselectivity, and the
diastereomers are not separable by column chromatography.
The oxazolidinone chiral auxiliary can be easily cleaved from the PK adducts (Scheme
5.53). Thus, the major diastereomer of
70a
(obtained in 66% yield) was dissolved in anhy-
drous allyl alcohol and heated at 150
◦
C in the presence of titanium tetraisopropoxide, to
afford the levorotatory allyl ester
72
(43% yield); on the other hand, the major diastereomer
of
70b
(obtained in 69% yield) was submitted to catalytic hydrogenation and reacted with
basic aqueous hydrogen peroxide, to furnish the ketoacid
73
in a 51% overall yield. The
circular dichroism of this compound established its absolute configuration.
24
(R = Ph)
O
O
Ph
R
2-propenol,
70a
(R = Ph)
70b
(R = SiMe
3
)
O
Cat. Ti(O
i
Pr)4
150 °C, 48 h
H
H
H
O
H
N
O
O
O
(Major isomer)
(-)-
72
Ph
H
2
, 10% Pd-C,
EtOH, rt, 6 h
(R = SiMe
3
)
O
O
LiOH, H
2
O
2,
THF, 0 °C, 2 h
73
O
H
H
H
H
H
OH
H
N
O
O
O
Ph
Scheme 5.53
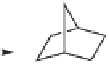
With the aid of semi-empirical PM3(tm) calculations, the authors rationalized the stere-
ochemical outcome of the reaction by means of the norbornadiene-dicobalt pentacarbonyl
complex depicted in Figure 5.1.
24
The olefin binds from its
exo-
face to an equatorial site
of the less-hindered pro-(
S
) cobalt, in an orientation that minimizes the steric interactions
of the methylene bridge with the alkyne substituents.
Hiroi and Watanabe investigated the use of (
S
)-methionine-derived amides as chiral
auxiliaries in the intermolecular PKR of phenylpropiolic amides with norbornene.
54
The
yields of the adducts were very variable, depending on the amide substituents. Reason-
able yields and diastereoselectivities were obtained in some instances (Scheme 5.54). The