Biomedical Engineering Reference
In-Depth Information
DNA strand, with bases numbered
5'
3'
1
2
3
4
5
6
7
8
9
10
Fragment generation via
chemical or enzymatic means
1
1
2
Seperation of each fragment
via electrophoresis
1
2
3
1
2
3
4
1
2
3
4
5
Etc.
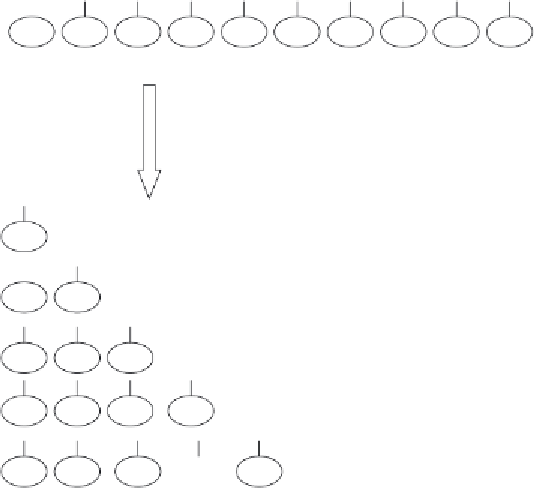
Figure 3.11
A simplifi ed overview of the approaches adopted to both chemical and enzyme-based DNA se-
quencing. Refer to text for details
3.3 Recombinant production of therapeutic proteins
The evaluation of any protein as a potential biopharmaceutical and its subsequent routine medi-
cal use are dependent upon the availability of suffi cient quantities of the target protein. In most
instances this is best achieved via production by recombinant means (i.e. via genetic engineering).
In addition to facilitating the production of any protein in substantial quantities, recombinant-
based production can have a number of additional advantages over direct extraction from a natu-
rally producing source, as described in Chapter 1. Production of any protein via rDNA technology
entails the initial identifi cation and isolation of a DNA sequence coding for the target protein.
This sequence can be direct genomic DNA, but mRNA coding for the protein of interest can also
act as a starting point. In the latter approach, the mRNA is enzymatically 'reverse transcribed'
into cDNA. If the target therapeutic protein is eukaryotic (which is invariably the case) then the
genomic DNA will contain both coding (exon) and non-coding (intron) sequences (Figure 3.9),
whereas the cDNA will be a refl ection of the exons only.
The desired gene/cDNA is normally amplifi ed, sequenced and then introduced into an expres-
sion vector that facilitates its introduction and expression (transcription and translation) in an
appropriate producer cell type. All recombinant therapeutic proteins approved to date are pro-
duced in
E. coli
,
S. cerevisiae
or in animal cell lines (mainly CHO or BHK cells). The general



Search WWH ::

Custom Search