Chemistry Reference
In-Depth Information
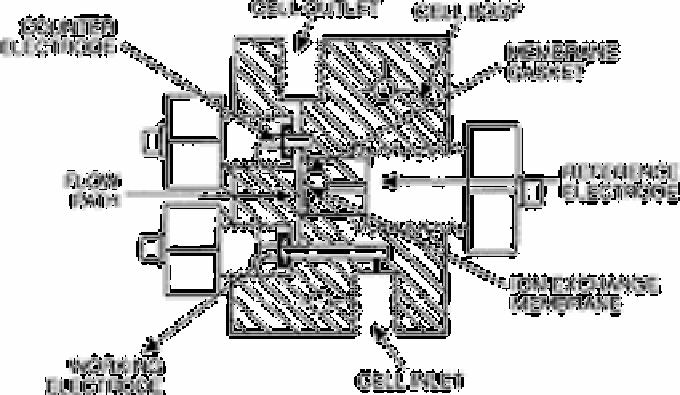
stream by a Nafion cation exchange membrane and platinum counter electrode. (Nafion
is a registered trade mark of E.I. du Pont de Nemours & Co.) The cell geometry is based
on one previously reported by Lown
et al.
[79]. The working electrode was occasionally
cleaned by mechanical
Fig. 12.37
Diagram of amperometric flow through cell
Source: Reproduced with permission from the American Chemical
Society [23]
polishing. The applied potential was 0.V for cyanide and sulphide, 0.20V for iodide, and
0.30V for bromide.
Fig. 12.38 shows the separation achieved on a 12 anion standard by this procedure.
Sulphide, cyanide, bromide, and sulphite are detected at the silver electrode while nitrite,
nitrate, phosphate and sulphate produce no response. Due to the low dissociation of
hydrogen sulphide and hydrogen cyanide following protonation by the suppressor
column, they are not detected by the conductivity detector.
The major advantage of ion chromatography over other analytical methods is its ability
to separate interferences. With one exception, the determination of one of the four ions is
not affected by the presence of the others. For example, a solution containing 2500 times
as much chloride as bromide has little effect on the determination of bromide (Table
12.30) even though chloride elutes first. Since E
0
for the oxidation of silver to silver
chloride is with respect to the value of E
0
for the oxidation of silver to silver bromide, at
a potential just on the diffusion controlled plateau for bromide, the current response for
bromide will be much greater than that for chloride. This can be exploited in order to
determine small quantities of bromide in a large excess of chloride, a difficult process
using other methods of analysis due to the similar chemical properties of the two halides.
In the determination of 50µg L
−1
bromide present in 1000mg L
−1
of chloride the large
negative dip following the chloride peak is caused by the reduction of the silver chloride
deposited on the electrode. Since there is no longer chloride in the solution next to the
Search WWH ::

Custom Search