Environmental Engineering Reference
In-Depth Information
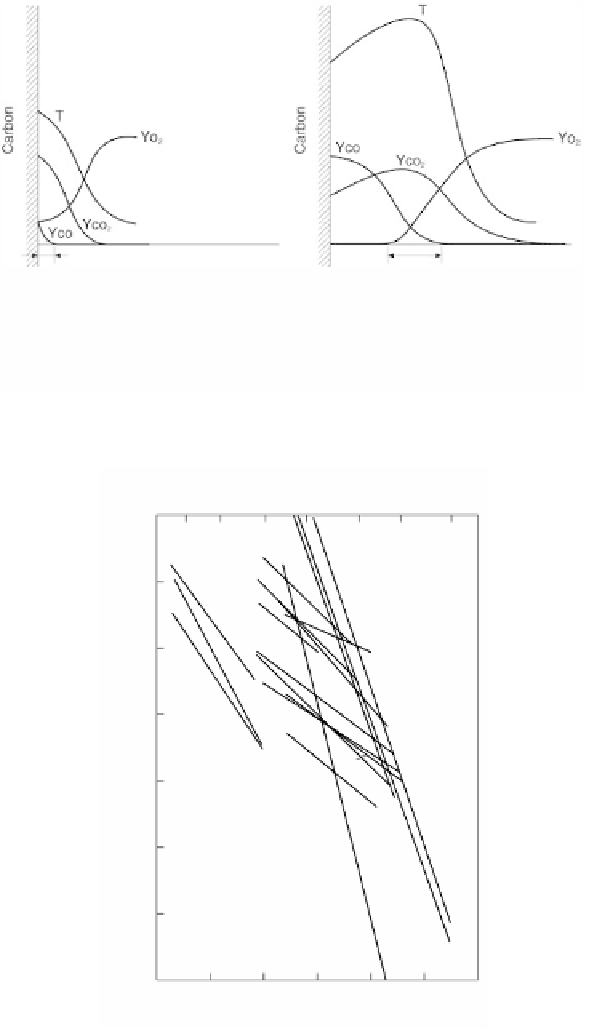
Gas-phase reaction zone
2CO
+
O
2
→
2CO
2
Surface reaction(i)C
(s)
+
O
2
→
CO
2
(ii)2C
(s)
+
O
2
→
2CO
(a) Case at low surface temperature
Gas-phase reaction zone
2CO
+
O
2
→
2CO
2
Surface reaction(iii)C
(s)
+
CO
2
→
2CO
In case (b), rates of
reaction (i) and (ii)
are less
(b) Case at high surface temperature
FIGURE 2.91
Combustion model of carbon.
Temperature, C
1800 1400
1000
800
600
500
400
10
3
1
e
10
a
5
b
5
a
2
10
2
14
18
a
10
1
12
1
a
1
b
1
c
10
0
18
b
4
a
6
a
10
-1
3
8
a
10
-2
6
b
10
-3
10
-4
0.4
0.6
0.8
1.0
1.2
1.4
1.6
Reciprocal absolute temperature, 10
-3
K
-1
FIGURE 2.92
Rate constant of emission process of volatile matter by the first-order reaction
model. (The numbers on the curves indicate types of coal and suffixes indicate combustion
condition.)

Search WWH ::

Custom Search